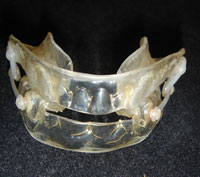
The mandibular protraction splint is a very effective treatment against snoring and sleep apnea syndromes.
A mandibular protraction splint is the only "sensible" treatment for excessive tongue volume or retrognathia in adults. It involves fitting a device that holds the mandible forward during sleep, which simultaneously advances the base of the tongue, giving it more space.
The mechanism of action of mandibular advancement operates on two levels:
• Increased diameter of the upper airways
• Reduced risk of upper airway closure due to tension in the pharyngeal wall
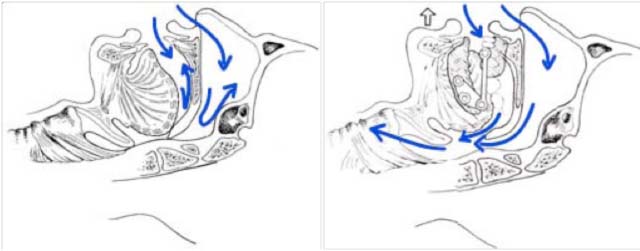
mechanism of action of orthotics
This treatment is very popular right now because it's simple and highly effective, but it's essential to be wary of the proliferation of orthotic models sold in pharmacies or online. To be effective, the orthotic must be custom-made after a dental assessment.
- Custom-made because it is thermoformed, it shifts at night and the propulsion is not suitable; a bipartite design is preferable as it is better tolerated and adaptable. Currently, only the ORM type mandibular protraction orthosis from RESMED/NARVAL laboratories is covered by social security under certain conditions.
- After a dental examination, because the traction forces are significant, it is necessary to verify the absence of tooth mobility, periodontal disease, and the solidity of the remaining teeth.
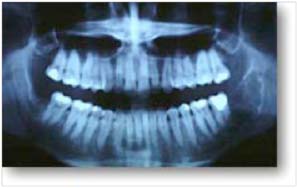
- A panoramic dental X-ray is often useful
WHAT TYPE OF ORTHOSIS SHOULD BE CONSIDERED?
There is a consensus in favor of custom-made bi-block orthoses.
- They are scientifically validated and recognized by the HAS (French National Authority for Health)
- Custom-made orthotics are more effective and better tolerated,
- Monobloc orthoses are being used less and less due to their lower tolerance; bibloc orthoses allow vertical and lateral movement and offer an advantage in terms of a lower risk of joint complications.
temporomandibular
Only custom-made bibloc orthoses produced by RESMED / NARVAL laboratories are recognized by the HAS and can benefit from reimbursement under conditions as specified in the official journal of October 31, 2008.
LPP Code: 2497884 Designation: MANDIBULAR ADVANCEMENT ORTHOSIS, NARVAL, ORM External orthoses and prostheses. Non-orthopedic external prostheses. ORM mandibular advancement orthosis, Narval Laboratories SA. Coverage for the ORM mandibular advancement orthosis is provided for the treatment of severe OSAHS (apnea-hypopnea index greater than 30 or, less than or equal to 30 and greater than or equal to 5 associated with severe daytime sleepiness) as a second-line treatment after refusal or intolerance of continuous positive airway pressure (CPAP) therapy. Coverage is provided after prior authorization completed by the prescribing physician for the initial prescription and each renewal. The response from the social security organization must be sent within the time limits stipulated in Article R. 16523 of the French Social Security Code. The ORM mandibular advancement device is a custom-made appliance. Prescribing this appliance requires collaboration between a sleep specialist (diagnosis, treatment, and follow-up) and a practitioner with expertise in both sleep and the masticatory system (dental examination, impressions, adjustments, and settings): – the diagnosis of sleep apnea must be documented by a clinical and polysomnographic examination (or by a respiratory polygraphy); – the prescription will be preceded by a dental examination to rule out any dental or joint contraindications. The effectiveness of the appliance must be monitored by a respiratory polygraphy or polysomnography. Rigorous long-term follow-up by a sleep specialist is required. A follow-up of the masticatory system is required every 6 months. Coverage for the ORM appliance excludes coverage for continuous positive airway pressure (CPAP) therapy. However, in the event of objectively proven failure of the appliance treatment, CPAP therapy may be offered. The orthosis is guaranteed for 1 year. Renewal is only authorized after a 2-year period following the previous fitting and is conditional upon: – demonstration of effectiveness (improvement of symptoms and a reduction of at least 50% in the AHI on the follow-up polysomnography under ORM); – adherence to the dental follow-up schedule. Any early renewal will require justification from the prescribing physician. Prior agreement: Yes
Official Journal Dates and Decree 31/10/2008 28/
ARE THERE ANY SIDE EFFECTS FROM WEARING AN ORTHOSIS?
- Short-term side effects are common but minor and generally transient (disappearing 10 to 30 minutes after waking up)
- Dental and gum pain
- Joint numbness and a sensation of loss of occlusion
- Hypersalivation
- Dry mouth
- The risks of orthodontic side effects are low, particularly when the advancement does not exceed 6mm.
- The main long-term risk (5 years and over) is incisor tipping due to the forces exerted by the groove in the anteroposterior direction.
- No triggering of temporomandibular joint pathology is observed, but rather an improvement, with the orthosis acting as a "stretching" effect and preventing teeth grinding (bruxism).
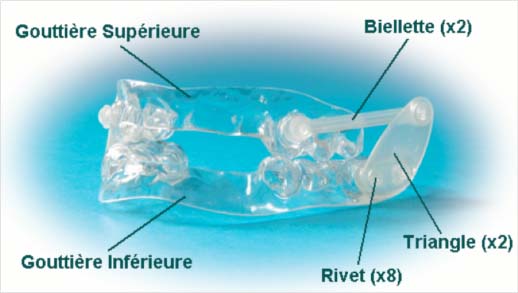
ORM traction NARVAL type orthosis
MONITORING OF ORTHOSIS TREATMENT
- A check of the effectiveness of the orthosis by nocturnal polygraphy is necessary in cases of sleep apnea
- Adapting mandibular propulsion by changing the size of the connecting rods according to tolerance and effectiveness is often necessary
- With regular use, the orthosis will need to be replaced after an average of 2 years